Novel Autophagy Pathway Links CD38 and LRRK2 to TFEB Nuclear Translocation
While autophagy is a well-known cellular process used to recycle cytoplasmic contents, the specific pathways and mechanisms of induction are still being elucidated and defined. A recent paper by Neel Nabar and colleagues from the National Institute of Health added new signaling molecules (CD38 and LRRK2) into the autophagy induction pathways.
Autophagy is a non-specific mechanism to degrade and recycle cytoplasmic material. The process begins with the sequestration of cytoplasmic material by a growing double membrane that matures into an autophagosome. The contents of the autophagosome are degraded after fusion with lysosomes and then recycled to the cytoplasm for reuse.1
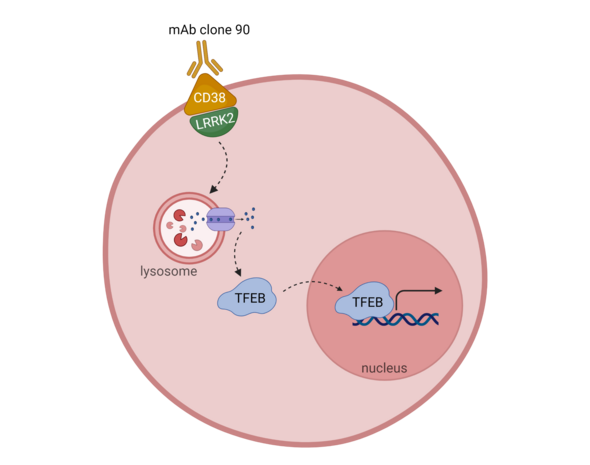
Transcription factor EB (TFEB) is a master regulator of autophagy affecting both transcription and protein levels. Autophagy is induced when TFEB translocates to the nucleus and increases expression of lysosomal biogenesis and autophagy genes. Nuclear translocation of TFEB is dependent on dephosphorylation, which can occur through a calcium dependent PP3/calcineurin mechanism.CD38 is a transmembrane protein containing multiple enzymatic domains that can function intracellularly or extracellularly to generate secondary messengers capable of calcium mobilization. Under acidic conditions, CD38 catalyzes the formation of nicotinic acid adenine dinucleotide phosphate (NAADP) from nicotinic adenine dinucleotide phosphate (NADP). This production of NAADP can mobilize calcium from the lysosome into the cytoplasm.2
LRRK2 (leucine-rich repeat kinase 2) is a multimeric protein containing multiple enzymatic (GTPase and kinase) and protein-protein interaction domains. LRRK2 cycles between the plasma membrane and the cytosol with different enzymatic activity levels based on its location. LRRK2’s complex structure implicates a variety of cellular functions including translational control, cytoskeleton formation, synaptic vesicle biology, and autophagy.3Since CD38 and LRRK2 have been shown to affect calcium mobilizing pathways and autophagy,2,3 Neel Nabar and colleagues from the National Institute of Health investigated whether these proteins contributed to the same autophagy pathway.4 The roles of CD38 and LRRK2 in autophagy were assessed using TFEB translocation to the nucleus as a marker for autophagy induction.
Activation of CD38 was induced by ligation with the monoclonal antibody (mAb) clone 90. This ligation resulted in endocytosis of CD38 and nuclear translocation of TFEB in bone marrow derived macrophages (BMDMs) and B cells. Calcium chelators, PP3/calcineurin inhibitors, and secondary messenger inhibitors reversed the TFEB nuclear translocation observed after ligation with mAb clone 90. Together these data show that TFEB nuclear translocation is a result of a NAADP-mediated lysosomal calcium response dependent on PP3/calcineurin signaling.
Adding to this story, Nabar et al. found that during unstimulated conditions CD38 and LRRK2 colocalize to the plasma membrane, but after ligation with mAb clone 90, the complex is internalized. Using LRRK2 deficient mice, LRRK2 kinase activity was shown to be required upstream of the NAADP signaling pathway essential for TFEB nuclear localization. Additionally, this activation was not due to defects in CD38 internalization or trafficking through the endosomal system.
While these data show CD38 and LRRK2 participate in the same pathway after ligation with mAb clone 90, expression of an LRRK2 kinase overactive mutant also induced TFEB nuclear translocation and increased transcription and expression of multiple autophagy genes in the absence of clone 90. In addition to activating TFEB, the LRRK2 kinase overactive mutant increased the stability of TFEB by increasing phosphorylation at the c-terminal end of the protein.
Nabar and colleagues elucidated a novel signaling pathway after CD38 ligation by mAb clone 90 suggesting the potential for additional CD38 ligands that can induce autophagy via calcium mobilization. This work provides a functional role for CD38 enzymatic activity that is not required shown to be important in other CD38 signaling pathways. While the role of LRRK2 is critical in this autophagy pathway, its specific function is still unknown, indicating more work still needs to be done. This novel CD38-LRRK2-TFEB signaling pathway adds to growing knowledge about the role of autophagy and immunity which will impact future work on health and disease.
Fortis Products Featured in the Article
References
- Mizushima N. Autophagy: process and function. Genes & Development. 2007;21(22):2861-2873. doi:10.1101/gad.1599207
- Malavasi F, Deaglio S, Funaro A, et al. Evolution and Function of the ADP Ribosyl Cyclase/CD38 Gene Family in Physiology and Pathology. Physiological Reviews. 2008;88(3):841-886. doi:10.1152/physrev.00035.2007
- Paisán-Ruiz C, Lewis PA, Singleton AB. LRRK2: Cause, Risk, and Mechanism. Journal of Parkinson’s Disease. 2013;3(2):85-103. doi:10.3233/JPD-130192
- Nabar NR, Heijjer CN, Shi CS, et al. LRRK2 is required for CD38-mediated NAADP-Ca 2+ signaling and the downstream activation of TFEB (transcription factor EB) in immune cells. Autophagy. 2022;18(1):204-222. doi:10.1080/15548627.2021.1954779